
Assessing the impacts of renewable energy developments on soil quality and health.
The following case story is provided by Dr. Helen Glanville from Keele University, UK. With help of Gasmet’s GT5000 Terra gas analyzer, researchers performed a sampling campaign to gain data on soil conditions on the site of the energy park.
Background
In September 2019, Keele University was granted planning permission to convert campus-based agricultural land to a renewable energy park. The development funded by energy company ENGIE will include 2 wind turbines, an industrial-sized battery to store generated electricity and 15,000 solar panels (Fig. 1). This is part of a pledge by the University to become Carbon neutral by 2030 and is part of the University’s new low carbon energy project.

Fig. 1: Solar panels and wind turbines (Source: Keele University)
Initially, construction was due to commence in early 2020 (Covid-19 put a halt to this with work only starting at the end of 2020/start of 2021). Given the original short timeframe, a group of researchers led by Dr. Helen Glanville quickly organized a sampling campaign for December 2019 to generate a baseline dataset of soil conditions before the start of the development. This sampling campaign was attempting to build a multi-layer map of the site from the subsurface through to the soil-atmosphere interface. The group of researchers included a multi-disciplinary team and included undergraduate and postgraduate students, Ph.D. students, technicians and research staff across many subject areas including Environmental Science, Geology, Physical Geography, Sustainability and Life Sciences. This new development provides a unique opportunity to create an environmental life cycle assessment of an “at-scale” renewables project on typical UK agricultural land, combining multi-disciplinary analysis including carbon, environmental and economic attributes from project inception to completion.
What we did: Our sampling campaign
Our initial sampling campaign commenced in mid-December 2019 when conditions were far from optimal, with the ground being frozen early in the morning (Fig. 2), then once thawed, the site became like a quagmire. However, we were determined to persevere knowing that we only had a narrow window to collect baseline data (If only we had known Covid would delay the start, we could have sampled at a warmer time of the year!).

Fig. 2: Frosty conditions on site (Image credit: Luke Hobson)
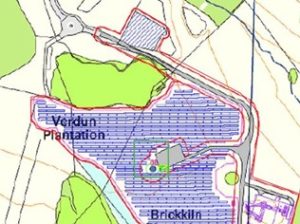
Given the size of the site, we opted to sample only a small 50 x 50 m2 area given our narrow time frame to collect data. We selected an area that would likely see a lot of activity (Fig. 3) and includes the location of one of the wind turbines, some solar panels, and the battery storage area. We selected a stratified random sampling approach to best to represent the selected area. Each point was located using real-time kinematic (RTK) global navigation satellite systems (GNSS) so we can go back during different stages of development and sample the same points.
 |
 |
Fig. 3. The left panel depicts the proposed development site with our sampling area highlight by the green box. The right panel portrays an aerial image of the site, boundary denoted by the red lines, sampling area denoted by green box and the red circles indicate our sampling points (Image credits: Ben Davenward)
What we measured and why: Soil quality and health
Soil quality is a measure of how well the soil can perform a particular function and often focuses on the physical, chemical and biological attributes of the soils such as soil structure, nutrient status, microbial activity, etc. (Fig 4) and is often a reflection of land management practices. In addition, soil health describes the resilience of the soil to adapt to change e.g., land-use change, climate stress, etc. In our study, we want to explore both aspects as the two are explicitly linked and both will likely be impacted through the development of the site.

Fig. 4. Dr. Helen Glanville and Dr. Adam Jeffery collecting soil samples to assess physical, chemical, and biological properties back in the laboratory (Image credit: Luke Hobson).
Sampling soil physical properties
Soil physical properties of the soil are critically important for sustaining life within the soil by influencing the ability of the soil to store essential water, air, and nutrients. Within this study, we took samples for bulk density, textural analysis, and soil water content. These measurements will help to understand the role of soil structure in influencing greenhouse emissions on this site. Also, we conducted a geophysical survey of the site using an EM-31 (electromagnetic conductivity) device to investigate, non-destructively, the subsurface (Fig. 5) including soil, groundwater features, shallow geology, or anything buried which may influence the conductivity of the area. This will also enable us to assess the impacts of construction on soil compaction, which in turn may have an impact on water holding capacity and could help to inform land management decisions around the site to improve soil quality.

Fig. 5. Ben Davenward and a postgraduate student undertaking an EM-31 geophysical survey to measure the subsurface electromagnetic conductivity (Image credit: Luke Hobson).
Sampling soil chemical properties
Soil chemical properties influence the biological activity within the soils as well as affecting plant growth above ground. The soil’s physical properties also strongly influence the soil nutrient status, so do the climate, aboveground vegetation and anthropogenic inputs or amendments. Here, we took soil samples and performed a series of chemical extractions to measure soil pH, EC, available nutrients (NO3, NH4, PO4), organic C and N both solid and dissolved forms, as well as measuring soil cation exchange capacity. We also installed rhizon samplers (Fig. 6) to directly access soil pore water in-situ, however, the near-zero temperatures made this very challenging as the majority of was locked up as ice and therefore unavailable. Finally, we conducted an in-situ soil elemental analysis using a Niton XL3 portable XRF analyzer capable of analyzing a whole suite of chemicals within the soil. This will also provide us with an opportunity to test the reliability of in-situ analysis compared to measuring elemental composition under controlled laboratory conditions both with the portable XRF as well as conducting full ICP-MS analysis.

Fig. 6. Rhizon sampling to extract soil pore water to analyze soil water nutrient chemistry (Image credit: Ben Davenward).
Sampling soil biological properties
Soils are living, breathing entities, and the combination of physical and chemical properties can influence the structure and composition of organisms that reside in the soil. Here, we conducted in-situ earthworm counts and species identification as well as collecting soils for microbial biomass determination and DNA sequencing. The construction will likely have an immediate impact on the soil biological communities initially, but what is of particular interest is how soil communities respond to the changes and whether we identify a shift in community structure as a result of the changing land use. Another avenue of interest is how potential vibrations from wind turbines may influence the earthworm behavior. Also, we are looking to work with the University Estates team and the construction company to explore ways to increase not only belowground biodiversity but also aboveground biodiversity on the site too, such as exploring planting regimes around the solar panels to encourage different pollinator species.
Soil organisms are also responsible for the emission of important greenhouse gases (notably CO2, N2O, CH4 and NH3). Soil disturbance is a major emitter of greenhouse gases and gives this site is to become a renewable energy park, we wanted to try and calculate the net loss of greenhouse gases before, during and after construction. It is hoped that by doing this, we can create an environmental life cycle assessment of “at-scale” renewables with a specific focus on soil emissions. To achieve this, we approached Ola Szymon at Gasmet to see if it would be possible for us to borrow the GT5000 Terra portable FTIR gas analyzer as we only could measure soil CO2, not any of the other important gases. To our delight, Ola was happy and willing to come help during our sampling campaign. This was a fantastic opportunity for us to see the kit in action (Fig. 7) and to see how quickly it took measurements of multiple gases simultaneously in-situ. The cold conditions did not affect its performance and we were successfully able to complete the full set of sample points in an afternoon.
|
|
 |
Fig. 7. Left: The Gasmet GT5000 Terra FTIR gas analyzer collecting greenhouse gas measurements. Right: Gasmet’s Ola Szymon and the Keele research team taking measurements (Image credits: Dr. Helen Glanville).
Despite our sampling campaign being conducted in the middle of Winter, with suboptimal conditions for measuring soil biological activity given the low temperatures, we identified variability in CO2 and CH4 measurements across our sampling area (Fig. 8, data only presented for CO2) as well as differences in soil conductivity readings indicative of changing soil water content across the site, which may be driving this variability.

Fig. 8. CO2 flux data from the Gasmet GT500 Terra FTIR analyzer across our sampling sites.
What next?
Our next steps are to work up the original dataset and create a multi-layer model of the site to look at the subsurface, surface and atmospheric interactions. Due to Covid-19 happening in early 2020, this has put a halt to our analysis due to not being allowed on campus and moving all our teaching to online delivery, which has taken a substantial amount of time. Our next steps include:
- Conducting a vegetation and biomass survey – ideally, we would like to conduct this during the plant growing season (March-September).
- Conducting small mammal surveys of the site to assess impacts on native species.
- Completing laboratory analysis for nutrient assays for baseline data
- Conducting additional multispectral and infrared thermal drone-based structure-from-motion photogrammetry survey to map soil moisture, vegetation, temperature, and topography.
- Conducting additional soil surveys to assess soil health throughout the development of the site.
- Generating a heat map to show spatial variability and interaction of characteristics across the site.
- Completing a full life-cycle assessment of an at-scale renewables project
- Exploring new land management practices on the development site to improve above and belowground biodiversity and to encourage pollinators.
- Designing and implementing new research experiments as part of our “living lab” to explore impacts of the solar panels and wind turbines on soil physical, chemical and biological properties.
Final remarks:
We would like to thank Ola and Gasmet for helping us to conduct this research at very short notice and for lending us the GT5000 Terra FTIR analyzer. We are also excited to announce that Keele University has recently secured UKRI World Class Laboratory funding to purchase our own gas analyzer. We have opted for the DX4015 as this has the additional capacity to cope with a humid environment which will enable us to not only sample terrestrial environments but also aquatic environments too. Hopefully, later this year, we will be able to return to campus and can continue this exciting avenue of research.

Fig. 9. Proof the sunshine did come out (at least once) (Image credits: Luke Hobson)
Interested in GHG monitoring technologies? Download our free guide to leading technologies to measure GHG flux from soil below:
SOIL FLUX MEASUREMENT REPORT
This study report compares six different technologies for measurements of greenhouse gases (GHGs) from the soil. Read the report and find out the agreement between different measurement technologies and suitabilities for GHG flux measurements from the soil!

